DESIGN
Only through a multidisciplinary approach can we obtain a range of products capableof responding to different anatomical, surgical, and prosthetic needs, while ensuringsafety and precision.
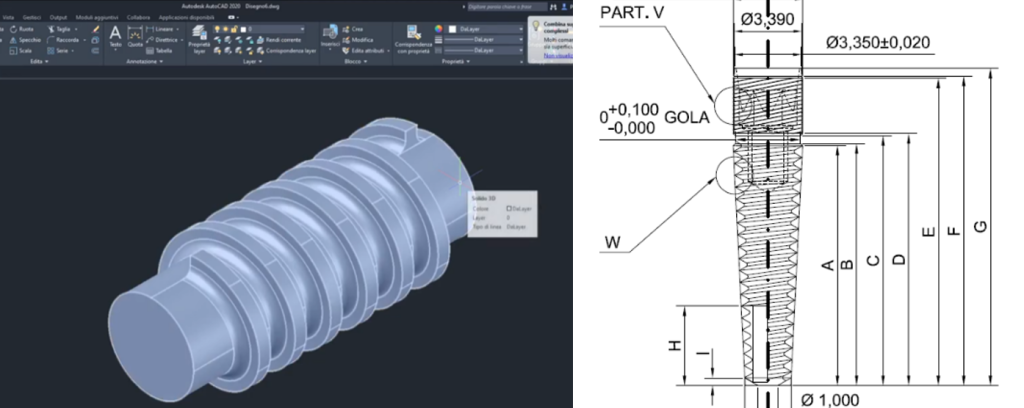
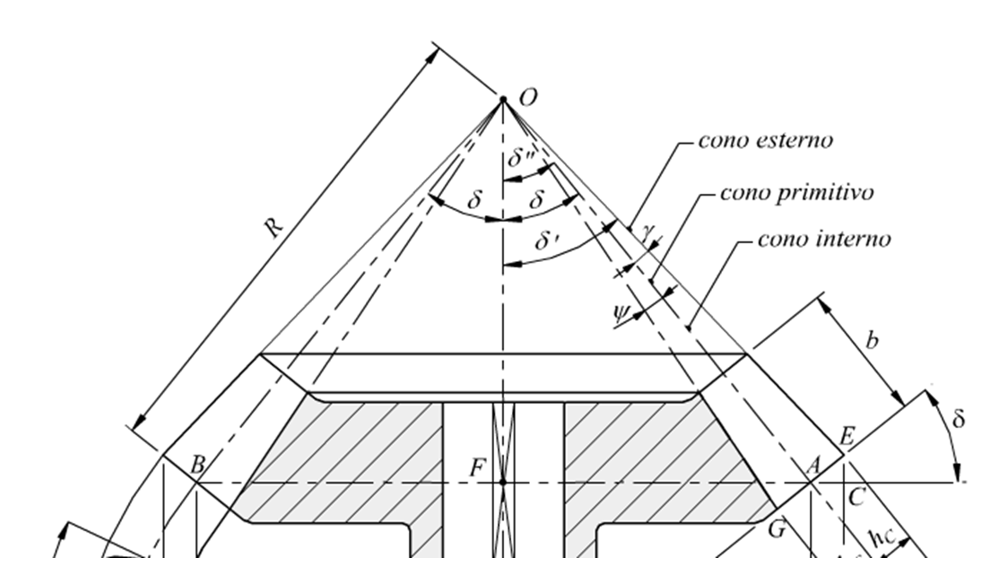
We use dedicated software to model the implant’s geometry.
• The following are defined: thread, internal/external connection, threads, prostheticplatform, etc.
• The design is conceived to ensure osseointegration and optimal distribution ofmasticatory loads.
• Technical tables are produced with orthogonal views, cross-sections, and millimetricdimensions with centesimal tolerances
. • Each component (screw, abutment, etc.) is represented with tolerances andproduction specifications.
PRODUCTION OF EXCELLENCE
MATERIAL SELECTION
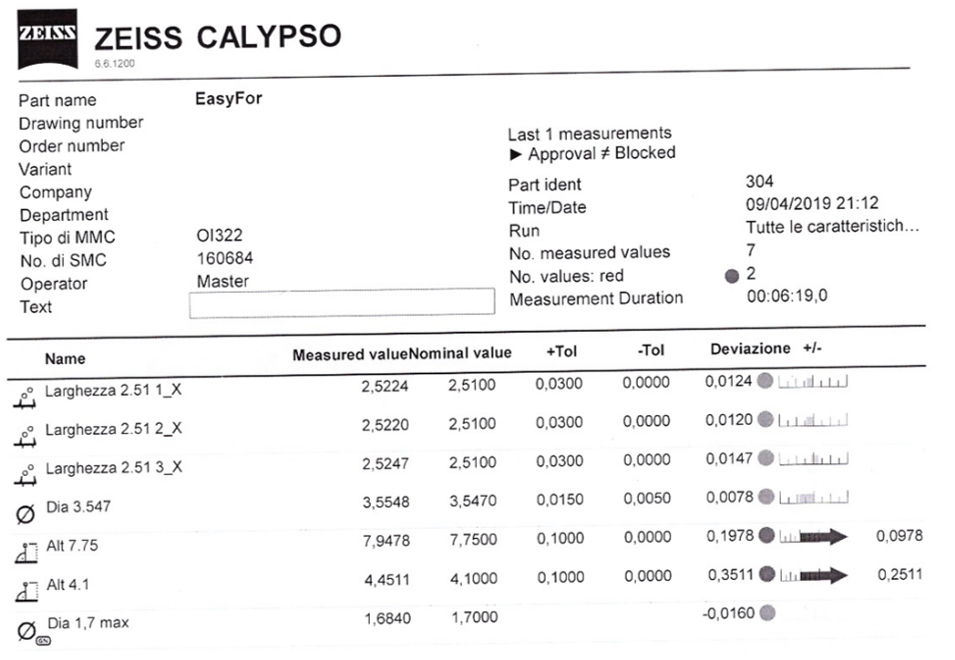
Strict Dimensional and Quality Controls
SURFACE TREATMENT AND DOCONTAMINATION
Treatment processes
• Sandblasting and double acid etching
• Final decontamination with plasma treatment
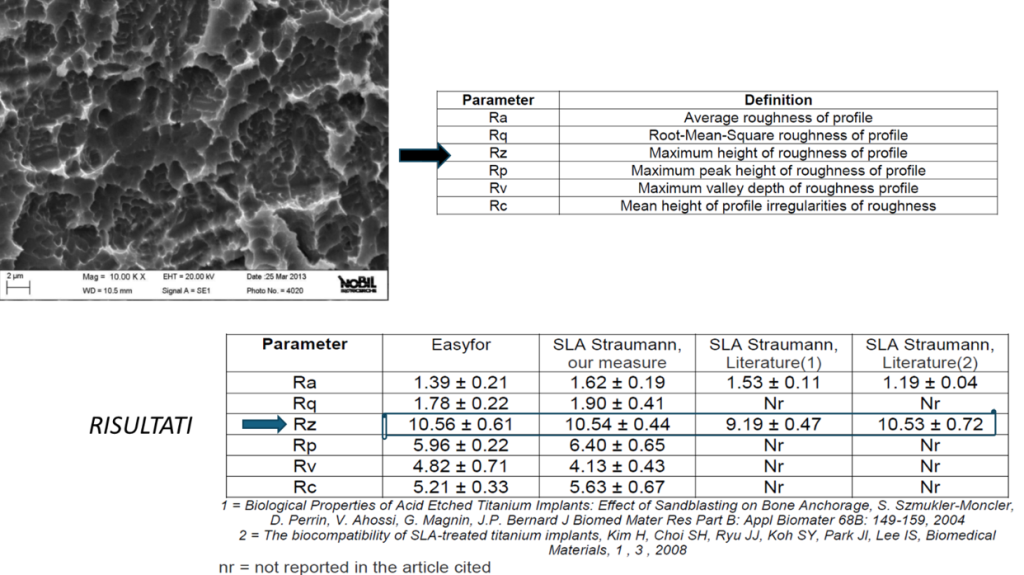
• Inspections and analyses
• Continuous monitoring of surface characteristics
Advanced characterization techniques:
• X-ray photoelectron spectroscopy (XPS)
• SemeDx
• AFM (Atomic Force Microscopy)
• Roughness measurements with StereoSEM
Complementary analyses:
• UV-VIS and infrared spectroscopy
PACKAGING
• Packaging in an ISO 7 clean room
• Sterile supply with Easyfor color code
• Double packaging:
– primary container consisting of a neutral POLYCARBONATE vial with a hermetic cap reproducing the color code
– secondary container consisting of a neutral POLYCARBONATE vial with a hermetic cap reproducing the color code with a titanium sleeve containing the implant
– insertion in a transparent medical polythene blister sealed by heat sealing with a label indicating the type of implant, diameter and length, in order to make identification of the product inside the package as simple as possible.
GAMMA RAY STERILIZATION
Easyfor entrusts its sterilization processes to Gammatom.
Gamma radiation is a high-energy electromagnetic wave characterized by deep penetration. Easyfor uses the radioisotope Cobalt-60, the only one used worldwide in photon irradiation processes.
• High penetration allows treatment on the final closed packaging
• No manipulation of products before, during, or after irradiation
• Stable environmental conditions (temperature, pressure, humidity)
• Absence of heat and differential pressures that could compromise the seal of the packaging
• No post-treatment residue
• DNA breakage with consequent impediment to cell replication
• Alteration of the structure of messenger RNA, no longer recognized by receptors
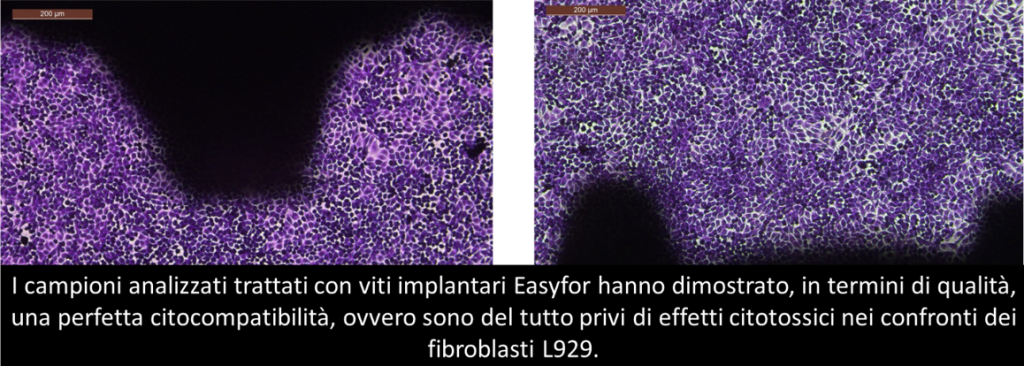
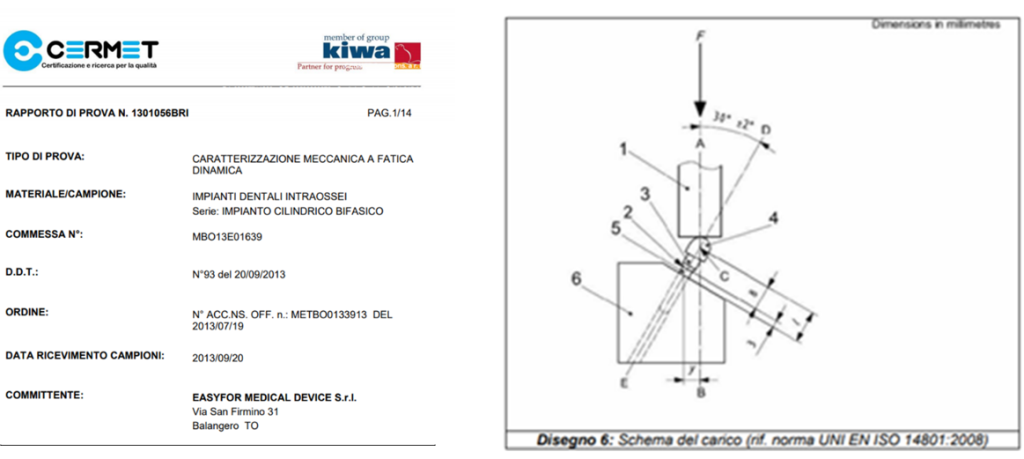
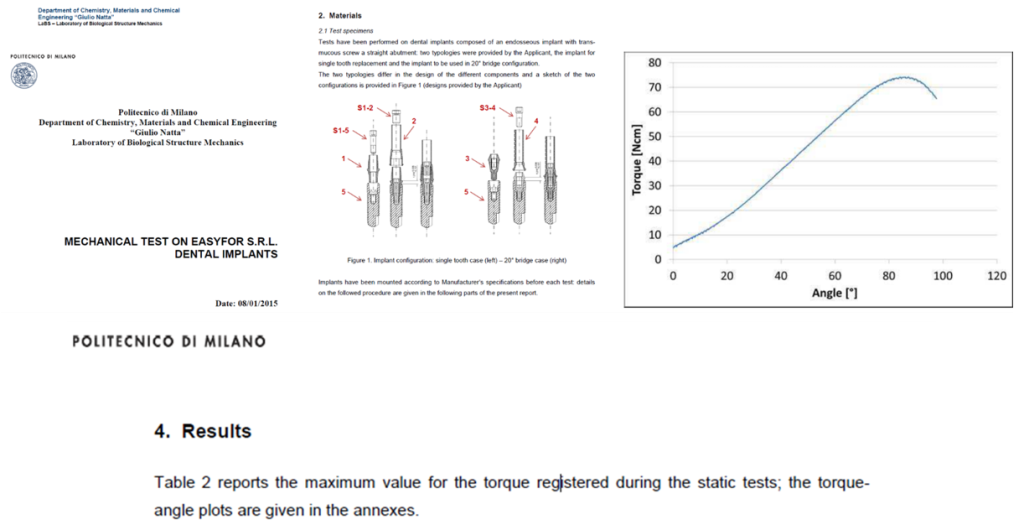
ELIGIBILITY CHECKS
Mechanical and stability tests
To ensure the durability and longevity of dental implants, Easyfor performs rigorous tests under simulated conditions of daily use.
They evaluate the implant’s resistance to:
• Occlusal forces
• Chewing cycles
• Mechanical torsion tests
To ensure robustness and reliability over time.
Long-Term Stability Testing
• Simulates implant performance under prolonged real-world conditions.
• Predicts durability, stability, and functionality for many years after placement.